Search
February 25, 2019
BioCorRx Appoints Two Finance Veterans to the Board of Directors
Page
ANAHEIM, CA - (NewMediaWire) - February 25, 2019 - BioCorRx Inc. (OTCQB: BICX) (“Company”), a developer and provider of advanced solutions in the treatment of substance use disorders, announced today the addition of two new independent directors, Louis Lucido and Luisa Ingargiola, to the Company’s Board of Directors.“We are delighted to welcome Lou and Luisa as new independent directors. Luisa has served as both a CFO and board member of multiple public companies, and has been instrumental in assisting several of these companies in the uplisting process to a senior national exchange. Lou brings extensive business acumen, having founded and helped run a multi-billion dollar asset management firm. As we continue executing on our growth strategy and plan for a Nasdaq listing, these new directors bring a wide range of relevant experience. We believe that the breadth, depth and diversity of their experience in both corporate finance and capital markets will strengthen BioCorRx on many levels,” said Brady Granier, CEO, President and Director of BioCorRx. “They have impressive track records and we look forward to working with them as we believe they will have a meaningful impact on BioCorRx’s future growth.”Louis LucidoLouis Lucido was formerly the Senior Advisor and Chief Operating Officer of DoubleLine Group, LP, a large investment firm with over $100 billion in assets under management. He recently retired in December 2018 and was one of the five founding partners. He was previously at TCW, where he served as a Group Managing Director. Prior to joining TCW in 2001, Mr. Lucido was the Chief Investment Officer for Delphi Financial Group (DFG) and was on several subsidiary boards. Before DFG, he was the Chief Operating Officer and Secretary for Hyperion Capital Management and was also a member of the Resolution Trust Advisory Committee. Since February 2013, he has served as a member of the Board of Directors of CASA of Los Angeles and is the current Chairman. Additionally, he was elected in 2013 and currently serves on the Boards of Junior Achievement, Southern California ,826LA and the Lupus Research Alliance (formerly the Alliance for Lupus Research). Mr. Lucido received his MBA in Management and Finance from New York University, and was a member of the Dean’s Advisory Board of the N.Y.U. Stern School of Business.Luisa IngargiolaLuisa Ingargiola presently serves as Chief Financial Officer of Avalon GloboCare, a leading global developer of cell-based technologies and therapeutics, where she helped navigate its Nasdaq uplisting in 2018. Luisa is a Board Director and Audit Chair of Electra Meccanica, a Nasdaq-listed company designing and manufacturing electric vehicles; she also serves on the board of Globe Photos, a leader in licensed sports photographic prints and iconic pop culture imagery; and she serves as director of Operation Transition Corporation, a strategic consulting and advisory firm that places ex-military special operations forces into corporate careers. Luisa holds a Bachelor of Science in Finance from Boston University, and an MBA in Health from the University of South Florida.About BioCorRxBioCorRx Inc. (OTCQB: BICX) is an addiction treatment company offering a unique approach to the treatment of substance abuse addiction. The BioCorRx® Recovery Program, a non-addictive, medication-assisted treatment (MAT) program, consists of two main components. The first component of the program consists of an outpatient implant procedure performed by a licensed physician. The implant delivers the non-addictive medicine, naltrexone, an opioid antagonist that can significantly reduce physical cravings for alcohol and opioids, and can prevent opioid overdose following relapse. The second component of the program developed by BioCorRx Inc. is a Cognitive Behavioral Therapy (CBT) program tailored specifically for the treatment of alcoholism and other substance abuse addictions for those receiving long-term naltrexone treatment. The Company also conducts R&D under its controlled subsidiary, BioCorRx Pharmaceuticals. For more ...
February 25, 2019
Smart Communications Turns to Huawei for 5G-Oriented DIS at HQ
Page
MAKATI, Philippines - (NewMediaWire) - February 25, 2019 - Before 2019 Mobile World Congress, Smart Communications announced that they are building the 5G oriented DIS (Digital Indoor System) in Smart Tower, to provide ubiquitous high-quality user experience while supporting smooth evolution to 5G. This is a significant step to embrace the arrival of upcoming 5G era.Smart Tower is a 36-story skyscraper in the heart of Makati CBD and is the headquarters of Smart Communications. Currently the indoor networks are supported by DAS which provides 2G, 3G and LTE services. SMART Tower houses thousands of Smart employees and are in constant need of data services to fulfill both their professional and personal needs throughout the day. With a highly tech savvy workforce, a Digital Indoor System provide the best network in capacity and user experience. As SMART Tower also houses the Innovation Lab focusing on next generation technology and use cases such as 5G, the DIS will ensure that when needed, the DIS will smoothly evolve to support 5G network supporting eMBB, uRLLC and eMTC use cases.The existing DAS networks have been strained to satisfy these service requirements. For example, DAS does not support smooth capacity expansion as reconstructions are required to introduce multi-antenna solutions, which is a key component to 5G. DAS also requires tremendous O&M efforts and takes a long time to construct, making it unsuitable to meet the requirements of fast deployment.As one of Huawei's DIS-based solutions, LampSite Pro is easy to deploy. It provides huge capacity, supports multi-mode multi-band applications, and E2E visualized O&M. These features give it unique strengths to tangibly improve coverage and eliminate blind holes in the offices scenario, helping provide ubiquitous MBB experience to end-users. Data obtained from a DIS deployment in one of the major entertainment arena demonstrated significant uplink and downlink traffic increment by 100+%, indicating an efficient release of long-suppressed traffic. The downlink rate at the user end was constant even with the increase of user traffic, with peak data rate of 300+Mbps recorded, showing a significantly improved user experience. Additionally, this digital indoor system has the capability to evolve into future-oriented 5G networks without the need for additional cabling.Joachim Horn, Chief Technology and Information Advisor of PLDT and Smart, said: “Boosting our indoor coverage is part of the broader network improvement program of PLDT and Smart, which is focused on creating the best customer experience especially for data. Improving indoor coverage is important because people use their data heavily while inside buildings.”John Leung John.leung@wmglobal.com
February 25, 2019
Huawei Helps LG Uplus Build a Gbps 5G Commercial Network, Enabling New 5G Business Applications
Page
Barcelona, SPAIN - (NewMediaWire) - February 25, 2019 - At the eve of 2019 Mobile World Congress, South Korean carrier LG Uplus and Huawei jointly announced the establishment of the Gbps 5G commercial network. The two sides have completed the deployment of more than 10,000 sites. LG Uplus and Huawei also cooperate to demonstrate the ultra-HD video and VR services based on 5G networks and enable new 5G commercial applications.At the LG booth in MWC2019, LG Uplus and Huawei will jointly demonstrate 5G network services. As an important application of eMBB, VR will become a mainstream service in the 5G era and has been widely recognized by global carriers. Cloud VR will become a killer application. On January 22, 2019, LG Uplus showcased the world's first 5G Cloud VR-based game. It fully leveraged the capabilities of 5G broad bandwidth and low latency, and won high praise from onsite media and customers.Huawei's 5G products are mature in commercial use and have been widely recognized in the industry. They support large-scale 5G commercial deployment in the world. On February 25, 2019, Huawei will release a full-scenario 5G commercial system to provide a simplified 5G solution, including network simplification, simplified architecture, simplified site, low power consumption, and simplified O&M. Huawei helps customers reduce deployment costs, support fast commercial deployment, and build the best 5G network. In Seoul, South Korea, LG Uplus collaborated with Huawei to deploy 5G base stations over 10,000 sites. Huawei 5G AAU is deployed on the network to provide continuous coverage of 5G network. The average data rate of Golden Cluster in Gangnam District exceeds 900Mbps, which provides customers with stable and high-performance. This also demonstrates the excellent performance of Huawei's 5G products.LG Uplus in South Korea and Huawei have been firmly engaged in 5G cooperation, jointly promoting the 5G commercialization process, and dedicated to providing the best mobile service experience for users.MWC 2019 will be held from February 25 to February 28 in Barcelona, Spain. Huawei will showcase its products and solutions at booth 1H50 in Fira Gran Via Hall 1, booth 3I30 in Hall 3, the Innovation City zone in Hall 4, booth 7C21 and 7C31 in Hall 7. For more information, please visit http://carrier.huawei.com/en/events/mwc2019John Leung John.leung@wmglobal.com
February 25, 2019
Cannabis Science Set to Integrate Its Sponsored Research In Nigeria to Include Its U.S. Patent Number 9,763,991 for Compositions of Cannabinol (CBN) for Treatment of Various Neurological Conditions
Page
Pre-Clinical Work and Animal Studies Underway, and Protocols for Clinical Studies Under Development Irvine, CA - (NewMediaWire) - February 25, 2019 - Cannabis Science, Inc. (OTC: CBIS), a U.S. company specializing in the development of cannabinoid-based medicines, announces that it intends to expand the scope of its sponsored research in Nigeria to include neurological conditions, including Post-Traumatic Stress Disorder (PTSD). CBIS and its research collaborator are currently investigating the potential beneficial effects of cannabidiol on oro-bucco-lingual dyskinesias, oxidative stress, and psychosis using animal studies and a clinical trial. CBIS recently announced receipt of U.S. Patent Number 9,763,991 for Compositions of Cannabinol (CBN) for Treatment of Various Neurobehavioral Disorders, Sleep Deprivation (Insomnia), Anxiety Disorders, and PTSD, among other target indications.The invention relates to a composition for use in the treatment of neurobehavioral disorders utilizing various Cannabis plant extracts comprising CBN preferably with other constituents of this plant for such use and a method for the extraction of plants. The plants or plant parts may for instance be derived from Cannabis Sativa and/or Cannabis Indica and/or Cannabis ruderalis and/or other elements, and mixtures thereof. The plant extracts derived showed particularly beneficial effects against Sleeping Disorders, in particular Insomnia, PTSD, and anxiety disorders including Attention Deficit Hyperactivity Disorder (ADHD).The animal studies are well underway and should be completed in the coming months. This study is important in a country like Nigeria where movement disorders are still common because the relatively inexpensive conventional antipsychotics are still widely in use. Movement disorders are known to be more common in patients on the conventional antipsychotics, though they have also been reported in the atypical antipsychotics. It is therefore pertinent to continue to search for new and better forms of management of this difficult to manage movement disorder in psychotic patients. CBIS has entered into an initial, one-year Research Collaboration Agreement with Stellenbosch University. CBIS and Stellenbosch University intend to jointly develop and investigate the use of Cannabinoids to treat chronic pelvic pain disorder/chronic prostatitis, and potentially other indications. Stellenbosch University (SU) is one of the oldest Universities in South Africa. The research is being conducted in South Africa and Nigeria.Cannabis Science is aggressively expanding the Company’s network of research centers. Negotiations are ongoing with several academic institutions in the United States and internationally to establish new, collaborative research agreements. These contracts will focus on several indications, and are expected to expand the research funded, and directed by the Company. In addition, Cannabis Science will develop multi-center, clinical trial networks with cannabinoid drug development.“We are encouraged with the preliminary results from our pre-clinical work and animal studies in Nigeria,” stated Mr. Raymond C. Dabney, CBIS’ President, CEO, and Co-founder. “The research may be of use in other movement disorders, such as Huntington’s Disease, Tourette’s Syndrome, Cerebral Palsy, and Dystonia. These could be related through the neurotransmitter, dopamine, modulated by the CB1 receptors. While we will continue our research in regard to oro-bucco-lingual dyskinesias, oxidative stress, and psychosis, we believe there is an opportunity to expand our research in Nigeria to include PTSD. Specifically, we will develop a protocol for a PTSD clinical study immediately and begin this study as soon as possible. This will allow the Company to leverage our patent for compositions of cannabinol (CBN), as well as our expertise with other cannabinoids.”“Our work in Nigeria, while independent of our research collaborations in the U.S., contributes significantly to the Global Consortium we launched last year,” added Mr. Dabney. The Cannabis ...
February 25, 2019
Aleafia Becomes First Cannabis Stock to Top TSX Venture 50 Performance Rankings -- CFN Media
Page
Seattle, WA - (NewMediaWire) - February 25, 2019 - CFN Media Group (“CFN Media”), the leading agency and financial media network dedicated to the North American cannabis industry, announces publication of an article discussing Aleafia Health Inc. (TSX-V: ALEF) (OTCQX: ALEAF) (FRA: ARAH). The vertically-integrated licensed producer with a clinic network and over 50,000 patients, was named as the 2019 TSX Venture 50 top performing company of the year. The TMX Group’s annual performance awards are based on three equally weight criteria, including market capitalization growth, share price appreciation and trading volume.The TSX Venture exchange has become a popular place for cannabis companies to list their shares, particularly after Canada legalized recreational cannabis last year, but 2019 was the first year that a cannabis company topped its annual performance rankings.The award demonstrates the company’s increasing popularity in the space. Over the past 52 weeks, the company’s stock price rose from a low of $0.47 to a high of $4.70 before settling at its current levels of around $2.37, making it a popular stock for active traders interested in the cannabis industry. As such, daily trading volume averaged roughly 1.42 million shares over the past three months, providing significant liquidity for investors.Research Proves a Strong CatalystAleafia Health’s stock reached all-time highs back in late-September when it partnered with Cronos Group Inc. to study the effects of medical cannabis on insomnia and daytime sleepiness. Under the terms of the deal, Cronos Group would provide funding for the study, which would be conducted by doctors at Aleafia’s health clinics. The company also formally launched Aleafia Labs to focus on these research projects.The company’s stock also moved sharply higher when it announced positive results from its benzodiazepine study. After conducting a study with 146 patients, the company reported that 45.2 percent of patients stopped consuming benzodiazepines—as a sedative or anti-anxiety medication—following a medical cannabis prescription. The patients further reported decreased daily distress due to their medical condition.These research projects leverage the company’s wholly-owned network of more than 20 Canabo Medical Clinics with over 50,000 patients. With the largest medical cannabis dataset in the world, the company enables researchers to validate cannabis science, product teams to innovate, and management to build a strong portfolio of intellectual property. The division had already generated $612,000 in revenue by the third quarter of 2018.Appealing to Both Traders & InvestorsMany cannabis stocks appeal to active traders that look for important catalysts and price movements, but only a fraction of these companies are also attractive on a fundamental basis to long-term investors. Aleafia Health is an existing licensed producer with an experienced management team, 98,000 kilograms per year of production capacity expected this year, and a network of medical cannabis clinics with over 50,000 patients.The company already generates tangible revenue from its medical cannabis clinics, research activities and supply agreement with CannTrust Holdings Inc.. After merging with Emblem Corp., the company has amassed a significant production footprint that it’s expanding further through strategic investments around the world. At the same time, its massive cannabis dataset provides an invaluable competitive advantage.Currently, the company trades with a market capitalization of about $375 million, which is lower than many other licensed producers. As the company scales up production and research activity, investors may become increasingly interested in the growing revenue and cash flow. The R&D activity could also pave the way towards valuable revenue streams from licensing, which may help boost revenue and margins over the long-term.Click Here to Download Aleafia Health Inc.’s Investor PresentationLooking AheadAleafia Health Inc. (TSX-V: ALEF) (OTCQX: ALEAF) (FRA: ARAH) represents a compelling ...
February 25, 2019
/C O R R E C T I O N -- eHealth, Inc./
Page
eHealth, Inc. (NASDAQ: EHTH), the nation's leading private online health insurance exchange, is correcting misstatements regarding non-GAAP net income and non-GAAP net income per diluted share for the ...
February 25, 2019
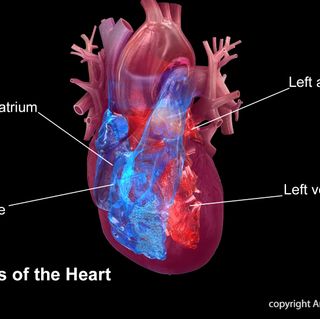
Better options needed for children at higher risk of premature heart disease
Page
Statement Highlights: New developments in identifying and treating the increased risk of premature heart disease in children and teens with certain medical conditions associated with increased cardiovascular risk are discussed in a scientific statement from the American Heart Association. Obesity and severe obesity are now considered significant risk factors for an increased risk of cardiovascular disease in children and teens. Embargoed until 4 a.m. CT / 5 a.m. ET Mon. Feb. 25, 2019 (NewMediaWire) - February 25, 2019 - DALLAS - Obesity and severe obesity in childhood and adolescence have been added to the list of conditions that put children and teens at increased risk for premature heart disease, according to a new scientific statement from the American Heart Association published in the Association’s journal Circulation. The statement provides an overview of current scientific knowledge about managing and treating the increased risk of atherosclerosis and early heart disease, in children and teens with type 1 or 2 diabetes, familial high cholesterol, congenital heart disease, childhood cancer survivorship and other conditions. Atherosclerosis is the slow narrowing of the arteries that underlies most heart diseases and stroke. “Parents need to know that some medical conditions raise the chances of premature heart disease, but we are learning more every day about how lifestyle changes and medical therapies that can lower their cardiovascular risk and help these children live their healthiest lives,” said Sarah de Ferranti, M.D., M.P.H., chair of the writing group for the statement and chief of the Division of Cardiology Outpatient Services at the Boston Children’s Hospital in Massachusetts. For example, there are treatments for familial high cholesterol – a group of genetic disorders that affect how people process cholesterol which can lead to extremely high cholesterol levels – that can help children and teens with this disorder live a normal lifespan. The statement is an update of a 2006 scientific statement and adds obesity and severe obesity to the list of conditions that put children and teens at increased risk of cardiovascular diseases and reviews new treatments for previously discussed conditions. Severe obesity and obesity are now considered moderate risk and at-risk conditions respectively because research shows they significantly increase chances of developing heart disease later in life. A study of almost 2.3 million individuals followed for over 40 years found the risks of dying from a cardiovascular disease were two to three times higher if their body weight as adolescents had been in the overweight or obese category compared to youth with normal weight. Effective treatments for obesity have proven elusive, but in general, a gradual approach to weight loss is generally required, incorporating improvements in dietary quality, fewer calories, more physical activity, meal replacements, medical therapy and/or bariatric surgery depending on the severity of the excess adiposity. Other significant changes to the statement since 2006 include: The elevation of Type 2 diabetes to a high-risk condition because of its association with additional cardiovascular risk factors such as high blood pressure and obesity. The expansion of the risks of premature heart disease associated with treatments for childhood cancers. Co-authors are Julia Steinberger, M.D., M.S.(Co-Chair); Rebecca Ameduri, M.D.; Annette Baker, R.N., M.S.N., C.P.N.P.; Holly Gooding, M.D., M.Sc.; Aaron S. Kelly, Ph.D.; Michele Mietus-Snyder, M.D.; Mark M. Mitsnefes, M.D., M.S.; Amy L. Peterson, M.D.; Julie St-Pierre, M.D., Ph.D.; Elaine M. Urbina, M.D., M.S.; Justin P. Zachariah, M.D., M.P.H.; and Ali N. Zaidi, M.D. Author disclosures are on the manuscript. Additional Resources: After Feb. 25, view the manuscript online. Obesity as young adult sets stage for heart disease, stroke Podcast - Children and Familial Hypercholesterolemia Age at cancer diagnosis may affect the risk of death from heart disease Follow AHA/ASA news on Twitter @HeartNews ### The ...
February 22, 2019
Zoom Telephonics Schedules Conference Call to Discuss Fourth Quarter and Year 2018 Financial Results
Page
Boston, MA - (NewMediaWire) - February 22, 2019 - Zoom Telephonics, Inc. (OTCQB: ZMTP) (“Zoom” or the “Company”), a leading producer of cable modems and other communication products, announced today that it expects to release its financial results for the fourth quarter and year ended December 31, 2018, after the close of market on Thursday, February 28, 2019. The Company will host a conference call at 4:30 p.m. Eastern Time that afternoon to discuss the results.Conference Call DetailsThursday, February 28, 2019 – 4:30 p.m. ETParticipant Dial-In Numbers:(United States): (866) 393-7958(International): (706) 643-5255Please dial-in five minutes prior to the start time of the call and provide the operator with the conference ID of 2280997.A slide presentation will accompany management’s remarks and will be accessible five minutes prior to the start of the call via the following link: www.zoomtel.com/SQ418. A recording of the call will also be made available afterwards through the investor information section of the Company’s website. About Zoom TelephonicsZoom Telephonics, Inc. designs, produces, markets, and supports cable modems and other communication products. The Company’s worldwide Motorola license agreement includes cable modems and gateways, DSL modems and gateways, cellular modems and routers and sensors, and other Internet and network products. For more information about Zoom and its products, please visit www.zoomtel.com/investor and www.motorolanetwork.com.MOTOROLA and the Stylized M Logo are trademarks or registered trademarks of Motorola Trademark Holdings, LLC and are used under license.Investor Relations Contact: Adam Prior, Senior Vice-President The Equity Group Inc. Phone: 212-836-9606 Email: aprior@equityny.com
February 22, 2019
Vodafone Spain Makes the First Standard 5G Call with Huawei Handset
Page
Madrid, Spain - (NewMediaWire) - February 22, 2019 - Vodafone Spain has become today the first operator in the world to make 5G standard calls from 5G mobile phones, after connecting the first smartphones to the 5G network with the specifications of the 3GPP NSA (Non Stand Alone) standard, which will be used later on commercial launch. The mobile phones have been connected to the Vodafone 5G network deployed in the center of Barcelona.The operator managed to implement various services, including several video calls to Spanish journalists via the commercial network between Barcelona and Madrid with a Huawei terminal. In mobile tests, download speed of up to 1.7Gbps were achieved. Both the smartphones used and the Vodafone 5G network meet the specifications of the Non Stand Alone 5G - 3GPP Release 15 standard, approved last year.Pilot projectVodafone already achieved in February 2018 the first 5G connection in the world between Castelldefels and Madrid, although on that occasion, the connection consisted of a videoconference between a PC connected to a 5G standard test network and a 4G mobile.It was also a pioneer in starting the deployment of a pre-commercial 5G network in Spain. In July 2018, it installed the first 5G NSA nodes in central areas of Madrid, Barcelona, Seville, Malaga, Bilbao and Valencia. This deployment began only days after acquiring 90 MHz of contiguous spectrum in the 3.7 GHz band.Vodafone Spain has already started several pilots in these cities to test possible 5G applications in the future. This is the case of the Connected Surgeon Project, in which a specialist surgeon guides, without being physically present and in real time, another surgeon who is operating in any operating room. Thanks to 5G technology, it is possible to transfer practical knowledge at critical moments. This pilot is part of a broad collaboration agreement that will make the Hospital Clínic de Barcelona the first 5G hospital in the country.John Leung John.leung@wmglobal.com